The clinical assessment of a 38-year-old female seeking preconception counseling necessitates a comprehensive evaluation of age-related reproductive dynamics. Advanced parental age serves as a critical determinant of fecundity, characterized by distinct physiological trajectories in males and females. Discrepancies in gametogenesis, neuroendocrine regulation, and the temporal rate of reproductive senescence yield significant implications for spontaneous conception and the efficacy of assisted reproductive technologies (ART). Furthermore, these dimorphic aging processes are intrinsically linked to adverse gestational outcomes and may modulate the long-term epigenetic and phenotypic health profiles of the progeny.
Female Reproductive Ageing
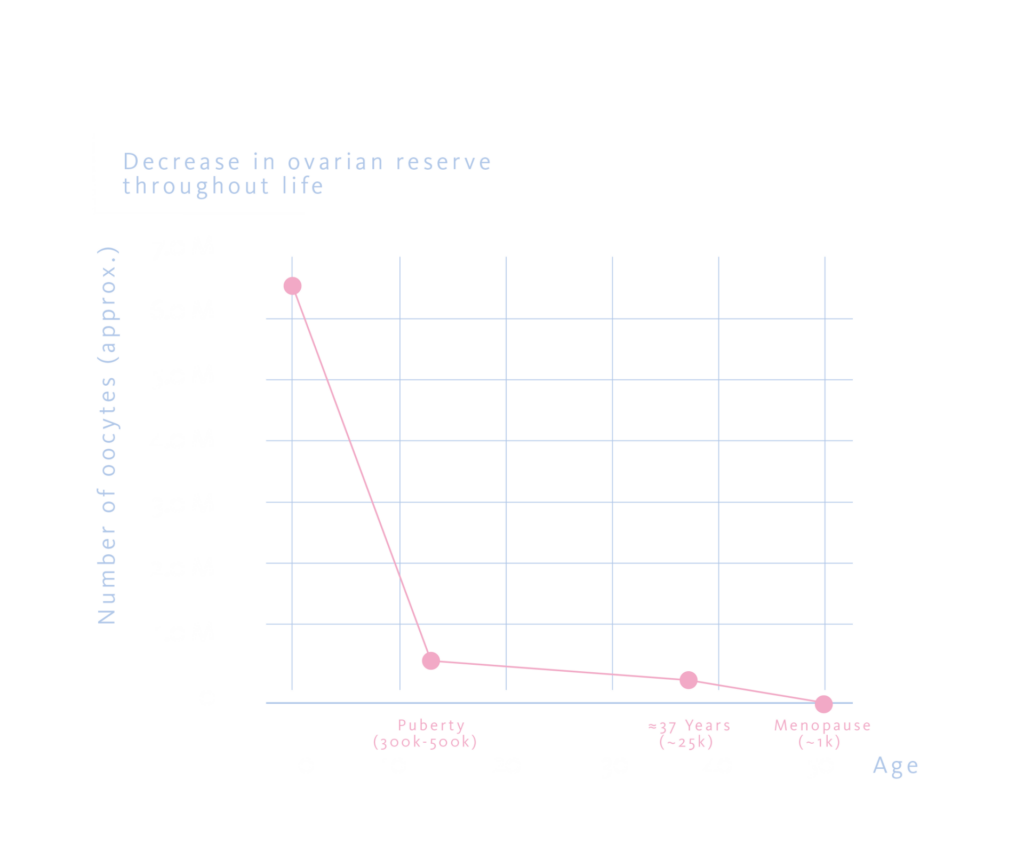
A woman’s reproductive potential is set before she is born, since her entire lifetime supply of oocytes is produced during foetal development and cannot be replenished later. The ovarian reserve reaches its highest point around mid‑gestation, when the foetus carries roughly six to seven million oocytes. By puberty, this declines to 300,000-500,000, and by age 37, only 25,000 remain. Menopause, typically around the early 50s, marks the end of natural reproductive capacity, leaving 1,000 residual oocytes.
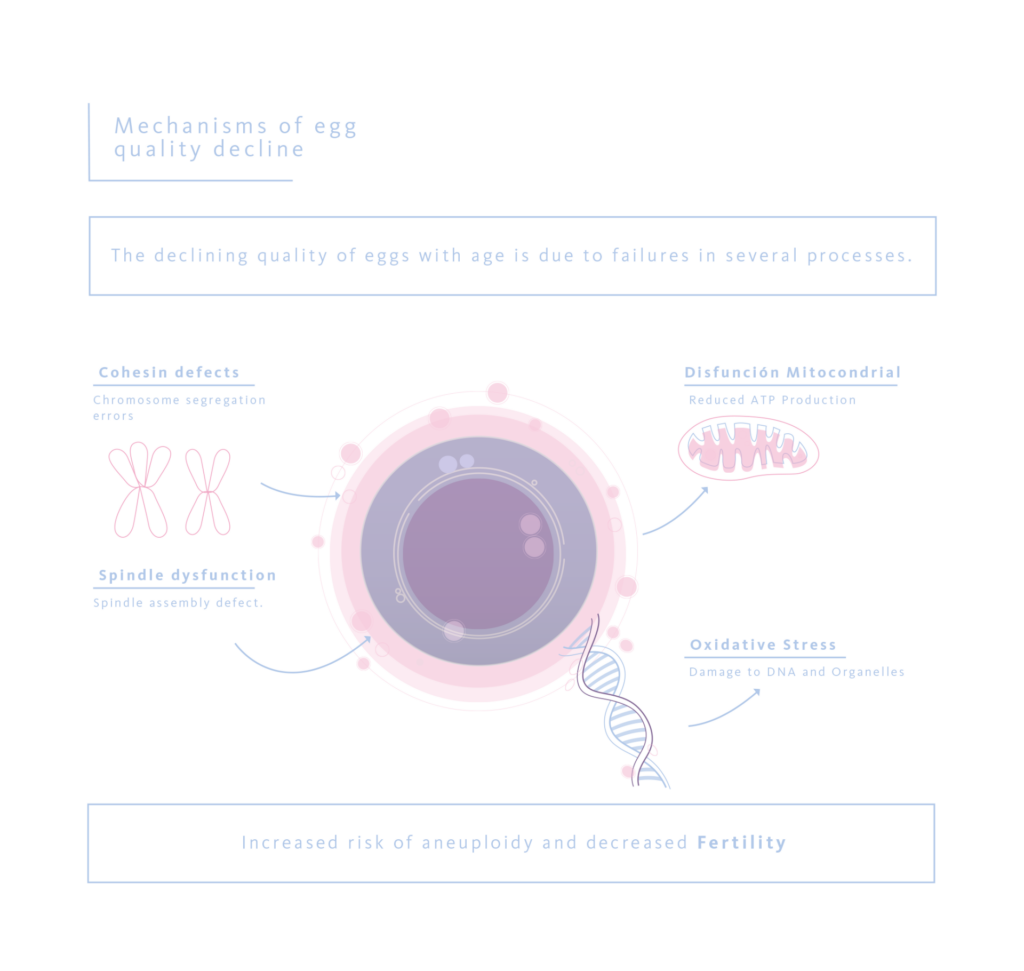
Mechanisms of Oocyte Quality Decline
A single defect does not drive the decline in oocyte quality with advancing age, but rather the progressive failure of several tightly regulated molecular and metabolic processes. One of the earliest changes involves deterioration of the cohesin complexes that maintain sister chromatid cohesion, a process essential for accurate chromosome segregation. As cohesin function declines, the risk of chromosomal missegregation increases. This defect is compounded by age-related dysfunction of the spindle assembly checkpoint, which reduces the oocyte’s ability to detect and correct misaligned chromosomes before meiotic progression.
Beyond these chromosomal control mechanisms, age-related alterations in cellular energetics play a central role. Aged oocytes show characteristic mitochondrial dysfunction, lower ATP output and alterations in NAD⁺-dependent metabolic pathways, which undermine the energetic demands of meiosis. In parallel, cumulative oxidative stress leads to the accumulation of reactive oxygen species, resulting in DNA damage and injury to key cellular structures. Together, these metabolic and oxidative injures further exacerbate the risk of aneuploidy and contribute to the progressive decline in oocyte competence observed with advancing maternal age.
All together, these changes offer a clear explanation for why the rates miscarriage, chromosomal abnormalities, and declining fertility tend to rise with maternal age, particularly from the late thirties onwards.
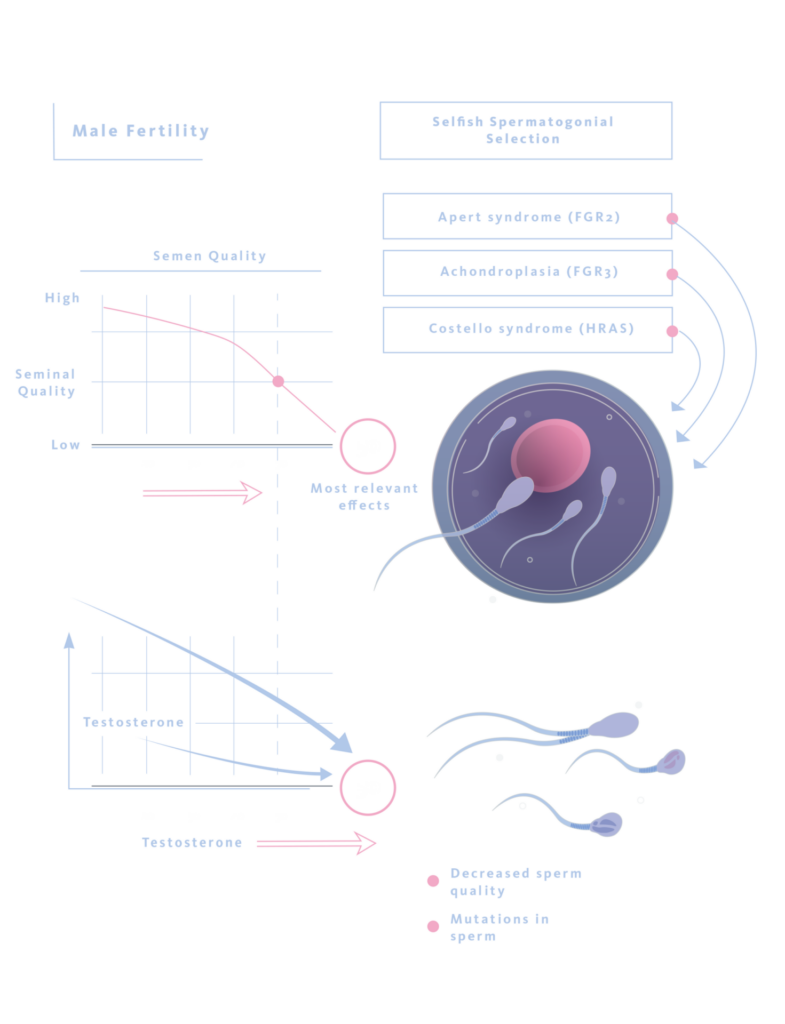
Male Reproductive Ageing
Although men continue to produce sperm throughout adult life, ageing is accompanied by slow but important changes in both sperm quality and hormonal balance. With increasing age, semen quality tends to deteriorate in several ways: sperm move less efficiently, DNA damage becomes more frequent, and new genetic mutations accumulate gradually, usually adding one or two each year.
Beyond the quantitative and structural changes observed in sperm with age, there is also a shift in the behaviour of spermatogonial stem cells. With time, certain de novo mutations appear to give a slight proliferative advantage to individual spermatogonial stem cells. As these mutated cells divided more frequently, they gradually occupy a larger proportion of the spermatogonial pool, a process known as selfish spermatogonial selection. This clonal expansion is thought to contribute to the increased incidence of several well-known paternal age-effect disorders. Well-described examples include activating mutations in FGFR2, associated with Apert syndrome, FGFR3, linked to achondroplasia, and HRAS, which causes Costello syndrome. Alongside these genetic changes, advancing paternal age is accompanied by epigenetic shifts in sperm. Changes in DNA methylation and histone marks can subtly reshape gene regulation in the early embryo, influencing developmental pathways in ways that go beyond the underlying DNA sequence. Ageing‑related shifts in the hormonal axis add an additional layer of complexity: testosterone levels tend to fall gradually with age, and the coordination of the hypothalamic-pituitary-testicular axis becomes less reliable over time. Clinically, these hormonal shifts are often reflected in a higher prevalence of erectile dysfunction and, indirectly, may contribute to less favourable reproductive outcomes
Measurable declines in semen parameters are typically observed after age 35-40, with clinically relevant fertility effects around age 50.
Paternal Age and Offspring Health
Large population cohorts consistently report a dose-dependent association between increasing paternal age and certain neurodevelopmental or psychiatric risks in offspring. Compared with fathers aged 20-24 years, those aged 45 years or older show significantly elevated risks across several neurodevelopmental and psychiatric outcomes. Estimates of the association between advancing paternal age and neurodevelopmental outcomes vary widely across studies and by condition. Some cohorts report moderately increased risks for disorders such as autism, ADHD or certain psychotic illnesses in the children of older fathers, while a few analyses have suggested larger effect sizes for specific diagnoses. These findings should be viewed with caution. Even when the relative risks reach statistical significance, the absolute risks for individual families remain low, and the implications for counselling should therefore be considered within the broader clinical and familial context.
Multigenerational studies indicate that older grand paternal age is also linearly associated with increased risks of schizophrenia, autism, ADHD, and intellectual disability, suggesting accumulation of de novo mutations across generations.
Fertility Evaluation and Clinical Guidelines
Current clinical guidelines recommend an age-stratified approach to fertility evaluation, reflecting the well-established relationship between advancing parental age and declining reproductive potential. In women under 35 years of age, formal assessment is generally advised after 12 months of regular unprotected intercourse without conception. For women aged 35 to 40 years, this interval is shortened to six months, recognising the more rapid decline in fecundity during this period. In women over 40 years, immediate fertility evaluation is recommended, given the sharply reduced likelihood of spontaneous conception and the time-sensitive nature of intervention.
In men, fertility assessment is typically initiated after 12 months of unsuccessful attempts at conception. However, earlier evaluation may be warranted in the presence of advanced paternal age or additional risk factors, such as known reproductive pathology, systemic illness, or prior gonadotoxic exposures.
These recommendations reflect the rapid, definitive female fertility declines and the more gradual male trajectory.
Assisted Reproductive Technologies (ART) and Age
Few areas of reproductive medicine illustrate the clinical impact of ageing as clearly as ART outcomes. Live birth rates per IVF cycle using autologous oocytes decline from 43% in women <35 to 3-4% in women >42. Cumulative success rates follow a similar trajectory. Donor oocyte cycles generally achieve 60-80% success, indicating oocyte quality is the primary limiting factor.
Notably, even with chromosomally normal donor oocytes, maternal age >45 may independently reduce live birth rates and affect perinatal outcomes, underscoring the contribution of uterine and systemic factors beyond embryo genetics.
Fertility Preservation
Several strategies are available to mitigate age-related reproductive decline, with approaches differing by sex. For women who are planning to delay childbearing, whether for personal, professional, or simply practical reasons, oocyte cryopreservation by vitrification has become the option most commonly recommended. In fact, the single factor that most strongly influences the likelihood of success is the woman’s age at the time the eggs are retrieved. As the years pass, both the number of available oocytes and their developmental competence decline in a gradual but steady way. Freezing eggs before 35-37 typically gives the best odds, but fertility varies from woman to woman. That’s why personalised advice is essential for each patient. Cumulative live‑birth rates per warming cycle are highest in women under 35 (approximately 49%), decrease through the late thirties, and fall markedly beyond age 40. Success is also closely linked to the number of mature oocytes stored. Modelling work suggests that storing roughly 15 to 20 mature oocytes gives a reasonable chance of achieving at least one live birth, especially for women in their mid- to late thirties. That said, these numbers come from population-level projections and shouldn’t be treated as individual forecasts, which is why personalised counselling remains essential.
In men, sperm cryopreservation remains the standard and well-established method for fertility preservation. It is widely used both for elective fertility preservation and before gonadotoxic treatments, offering a reliable means of safeguarding future reproductive potential.
Conclusion and Future Directions
Reproductive ageing unfolds differently in women and men. For women, fertility often declines over a relatively short period and once lost, cannot be regained. In men, it unfolds more slowly but is still clinically meaningful. Advancing maternal age affects both oocyte competence and endometrial receptivity, while increasing paternal age is associated with a growing burden of de novo mutations, the expansion of particular spermatogonial clones, and age-related epigenetic changes. Importantly, these influences may extend across generations, highlighting their wider relevance for offspring’s health.
Clinicians need to appreciate how factors like cohesion deterioration, mitochondrial impairment, and oxidative stress contribute to reproductive ageing. Understanding these mechanisms allows them to provide couples with practical, evidence-based guidance rather than abstract statistics. Several emerging interventions, including strategies aimed at supporting mitochondrial function or NAD+-dependent metabolic pathways, remain experimental and are currently backed only by limited and heterogeneous human evidence. Nevertheless, these approaches may represent future avenues to preserve oocyte competence and mitigate age-related decline, underscoring the ongoing evolution of this field.
As our understanding of these mechanisms deepens, the challenge for clinicians will be translating molecular insights into actionable guidance for the couples sitting across from them in the consultation room.
References
- Aitken RJ. Male reproductive ageing: a radical road to ruin. Hum Reprod. 2023 Oct 3;38(10):1861-1871. doi: 10.1093/humrep/dead157. PMID: 37568254; PMCID: PMC10546083.
- Bhasin S, Kerr C, et al. The Implications of Reproductive Aging for the Health, Vitality, and Economic Welfare of Human Societies. J Clin Endocrinol Metab. 2019 Sep 1;104(9):3821-3825. doi: 10.1210/jc.2019-00315. PMID: 30990518; PMCID: PMC11651520.
- Boxem AJ, Blaauwendraad SM, et al. Age among women and men, time to pregnancy and risk of miscarriage. BMC Med. 2025 Nov 17;23(1):639. doi: 10.1186/s12916-025-04462-8. PMID: 41250147; PMCID: PMC12625503.
- Ghazi A, Henry H. Reproductive Fitness and the Links to Chronic Disease and Systemic Aging. Physiology (Bethesda). 2025 Oct 21:10.1152/physiol.00013.2025. doi: 10.1152/physiol.00013.2025. Epub ahead of print. PMID: 41117435; PMCID: PMC12566053.
- Mahajan NK. Optimizing advice and approaches for elective fertility preservation. Best Pract Res Clin Obstet Gynaecol. 2025 Mar;99:102591. doi: 10.1016/j.bpobgyn.2025.102591. Epub 2025 Mar 14. PMID: 40106848.
- Zweifel JE, Woodward JT. The risky business of advanced paternal age: neurodevelopmental and psychosocial implications for children of older fathers. Fertil Steril. 2022 Dec;118(6):1013-1021. doi: 10.1016/j.fertnstert.2022.10.029. Epub 2022 Nov 5. PMID: 36347660.
