Safety of the donors and quality of the oocytes: The key to success
Infertility is a global health issue affecting millions of couples worldwide. Assisted reproductive technology (ART) has emerged as a key intervention for addressing various forms of infertility, with egg donation being an integral component of such treatments, specifically for individuals with diminished ovarian reserve or genetic concerns. Over the past 20 years, there has been a steady increase in the demand for gamete donation, and in Europe alone, in 2019, 82.373 egg donation cycles were performed (Gliozheni et al., 2023).
Given the increasing demand for egg donation and the greater complexity of this system, finding the best strategy to optimize controlled ovarian hyperstimulation (COH) in terms of donor safety and egg retrieval efficiency on one hand, and the treatment’s effectiveness in the recipient patient on the other, is of crucial importance.
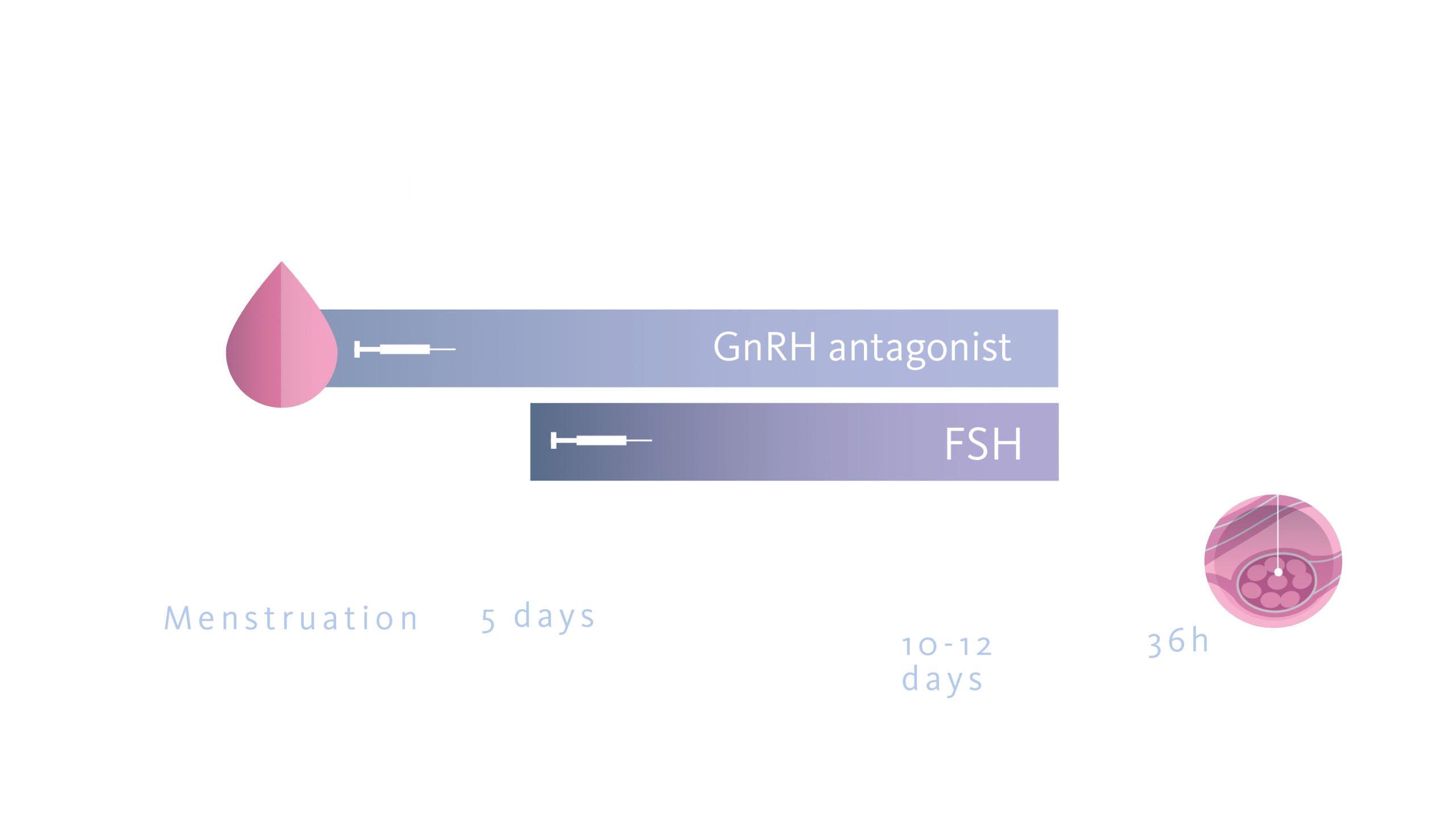
Over the past two decades, there has been a continuous evolution in COH protocols to maximize oocyte yield while minimizing risks, particularly the occurrence of ovarian hyperstimulation syndrome (OHSS). In particular, the advent of the short protocol with GnRH antagonist and the more recent progestin-primed ovarian stimulation protocol (PPOS), in conjunction with the trigger with GnRH agonist, has increased the safety of the procedures (ESHRE Ovarian Stimulation Guideline Development Group, 2019) (Martinez et al., 2021). The combined oral contraceptive pill (COCP), traditionally employed for menstrual cycle synchronization, presents both facilitative and potentially inhibitive effects on ART outcomes. The rationale for its use includes scheduled cycle starts and potentially enhanced follicular cohort uniformity but uncertainty prevails regarding its impact on the efficiency of subsequent oocyte retrieval and live birth rates (LBR).
Pill or not Pill: controversies of the use of oral contraceptives in ovarian stimulation cycles
The combined oral contraceptive pill (COCP) leads to long-term pituitary suppression and a reduction in LH levels in the early follicular phase, which could reduce the ability to effectively respond to the GnRH agonist trigger (standard in egg donation cycles) (Martinez et al., 2021). In fact, for this drug to be effective, the pituitary gland must be susceptible to stimulation by GnRH agonists. If the pituitary is not sensitized or unable to produce LH, oocyte maturation does not occur, resulting in lower oocyte yield, defined as the ratio of mature oocytes obtained to ovarian follicles present on the day of the trigger, than expected (Popovic-Todorovic et al., 2019). (Meyer et al., 2015).
Furthermore, we know that two important predictive factors of ovarian functional reserve and, therefore, the response to ovarian stimulation, are represented by serum levels of Anti-Mullerian Hormone (AMH) and the antral follicles count (AFC), as emphasized in the latest ESHRE guidelines on ovarian stimulation (Strong Recommendation) (ESHRE Ovarian Stimulation Guideline Development Group, 2019) and reiterated in a recent meta-analysis (Martinez et al., 2021). It has been demonstrated that therapy with COCP and/or strong progestins can cause a reduction in serum AMH levels (David H. Barad, 2021) and the number of antral follicles, both reversible.
Despite this evidence, few studies have been conducted to evaluate the effect of COCP in egg donation cycles, which have shown controversial effects (Barad et al., 2013) (Llácer et al., 2011) on the number and quality of eggs and, secondarily, on pregnancy outcomes in the recipients. Furthermore, all studies conducted so far have considered short protocols with GnRH antagonists or long protocols with GnRH agonists, while none have evaluated the effects in the case of using a PPOS protocol for pituitary suppression. On the other hand, it should be noted that in the latest international guidelines on COH, the use of pretreatment with COCP (even for 12-28 days) in protocols with GnRH antagonists has been strongly discouraged (Strong Recommendation) because the efficacy of such cycles would be reduced (ESHRE Ovarian Stimulation Guideline Development Group, 2019). The same guidelines conclude that, to date, there is no data to determine whether the negative effect of COCP also exists in different patients’ population (such as oocyte donors) and when other types of protocols, such as the PPOS protocol for pituitary suppression, are used.
To clarify these uncertainties, this multicenter retrospective cohort study assessed the outcomes associated with COCP synchronization in ovarian stimulation protocols involving progestin for pituitary suppression in egg donation treatments. Our analysis focused on the evaluation of mature oocyte yields among donors and pregnancy rates among recipients, aiming to discern the pragmatic implications of COCP pretreatment in a PPOS context for enhancing clinical regimens in ART.
Study design, results & conclusions: answering current doubts
Multicenter retrospective cohort study evaluating outcomes of egg donation cycles with and without COCP pre-treatment. A comparative retrospective cohort study (Analysis A) was conducted on 397 COH cycles (290 with COCP and 107 without COCP), and subsequently, a comparative paired study (Analysis B) was performed by extrapolating donors undergoing at least two cycles (one with and one without EP: 90 cycles), in which, to attempt to eliminate possible bias due to interpersonal differences, every patient was her own control. The retrieved oocytes were used for 216 egg donation cycles performed at a private clinic (gamete-providing clinic) and a public hospital (transnational egg donation program). Donors were assessed for the number of total, mature MII, and useful oocytes (without intra- or extracellular dysmorphisms suitable for ICSI), total FSH dose, days of COH, and cancellation rates (for low response or low serum LH on the first day of COH), while recipients were assessed for ongoing pregnancy rates (OPR), live birth rates (LBR), fertilization rates (FR), blastocyst formation rates (BR), embryo implantation rates (IR), and miscarriage rates (MR).
In donors, the “COCP-NO” group obtained a higher number of total, MII, and useful oocytes (statistically significant difference in comparative Analysis B), and similar stimulation days, total FSH dose used, and cancellation rate due to low response, while the “COCP-YES” group showed a significantly higher percentage of cancellations due to low LH levels, indicative of long-lasting pituitary suppression COCP related. In recipients, the “COCP-NO” and “COCP-YES” groups showed similar FR and BR, although the percentage of usable blastocysts was significantly lower in the ” COCP -YES” group when vitrified oocytes were used. IR was higher in the ” COCP -NO” group. MR was significantly higher in the COCP -YES group, while higher OPR and LBR were obtained in the COCP -NO group, with significant differences in the total population and in the vitrified oocyte subgroup.

Donor response to COH with PPOS protocol appears to be negatively influenced by COCP synchronization, with implications for egg donation treatment outcomes in recipients, including higher MR rate, lower OPR/LBR. This could be extended to fertility preservation programs in normo-responders patients, in which the probability of future pregnancy is closely linked to the number of good oocytes vitrified. Further studies are needed to confirm these findings.
References
- Gliozheni, O., Hambartsoumian, E., Strohmer, H., Petrovskaya, E., Tishkevich, O., De Neubourg, D., Bogaerts, K., Balic, D., Antonova, I., Cvetkova, E., Rezabek, K., Kirk, J., Sõritsa, D., Gissler, M., Pelkonen, S., Mansouri, I., de Mouzon, J., Tandler-Schneider, A., Kimmel, M., … Goossens, V. (2023). ART in Europe, 2019: results generated from European registries by ESHRE. Human Reproduction, 38(12), 2321–2338. https://doi.org/10.1093/humrep/dead197
- Martinez, F., Racca, A., Rodríguez, I., & Polyzos, N. P. (2021). Ovarian stimulation for oocyte donation: a systematic review and meta-analysis. Human Reproduction Update, 1–24. https://doi.org/10.1093/humupd/dmab008
- Popovic-Todorovic, B., Santos-Ribeiro, S., Drakopoulos, P., De Vos, M., Racca, A., MacKens, S., Thorrez, Y., Verheyen, G., Tournaye, H., Quintero, L., & Blockeel, C. (2019). Predicting suboptimal oocyte yield following GnRH agonist trigger by measuring serum LH at the start of ovarian stimulation. Human Reproduction, 34(10), 2027–2035. https://doi.org/10.1093/humrep/dez132
- Meyer, L., Murphy, L. A., Gumer, A., Reichman, D. E., Rosenwaks, Z., & Cholst, I. N. (2015). Risk factors for a suboptimal response to gonadotropin-releasing hormone agonist trigger during in vitro fertilization cycles. Fertility and Sterility, 104(3), 637-642. https://doi.org/10.1016/j.fertnstert.2015.06.011
- ESHRE Ovarian Stimulation Guideline Development Group. (2019). ESHRE Guidelines: Ovarian Stimulation for IVF/ICSI. Human Reproduction Open, October, 1–136.
- David H. Barad, M. D. , M. S. (2021). Antimullerian hormone–levels not written in stone. Https://Www.Fertstertdialog.Com/Posts/32990, 116.
- Barad, D. H., Kim, A., Kubba, H., Weghofer, A., & Gleicher, N. (2013). Does hormonal contraception prior to in vitro fertilization (IVF) negatively affect oocyte yields? – A pilot study. Reproductive Biology and Endocrinology, 11:28, 1–6. https://doi.org/10.1186/1477-7827-11-28
- Llácer, J., Moliner, B., Gimenez, J., Luque, L., Ten, J., & Bernabeu, R. (2011). Oral contraceptive pill (OCP) pretreatment in donors has not detrimental impact in egg donation treatments. Fertility and Sterility, 96(3), S176–S177. https://doi.org/10.1016/j.fertnstert.2011.07.688
